Having information about brain volumes at one-point-in-time can be a powerful tool in the evaluation of neurodegenerative diseases. However, personalized patient care does not end after one time point. It is an ongoing process that includes continuous monitoring, evaluation of care effectiveness, and potentially, adjustment to previous care decisions.
Volumetric brain measurements provide a great deal of valuable information to a physician, especially after an incident, or after a change in treatment. These are some of the questions that could be answered by a trained physician using the NeuroQuant® longitudinal reporting feature.
- Is the brain volume deterioration continuing, may indicate that the treatment is not as effective as expected?
- In dementia patients: Are the patient’s memory complaints from normal aging or has brain atrophy progressed?
- In multiple sclerosis patients: Has the brain volume loss stopped? Is there increased damage from recent episodes?
- Is the current treatment plan effective in slowing or stopping further neurodegeneration?
Longitudinal report case study:
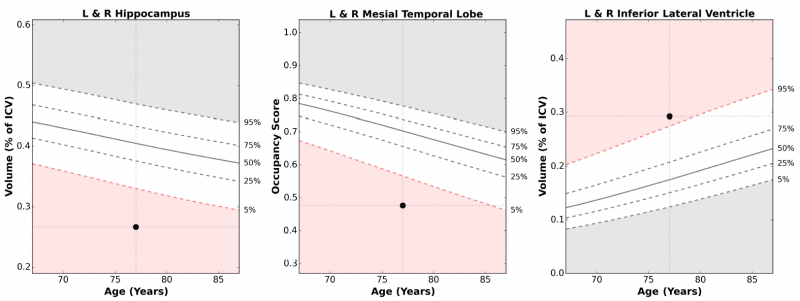
The chart below shows the NeuroQuant results for a 77 year old patient with memory loss complaints. His hippocampal volume is smaller than expected for when normalized to age and gender and outside the 95 percentile (left graph), the inferior lateral ventricles are larger than expected when normalized for age and gender and outside the 95 percentile (right graph) and the hippocampal occupancy score (HOC) of the mesial temporal lobe is also outside the 95 percentile. These results show strong indications of hippocampal atrophy.
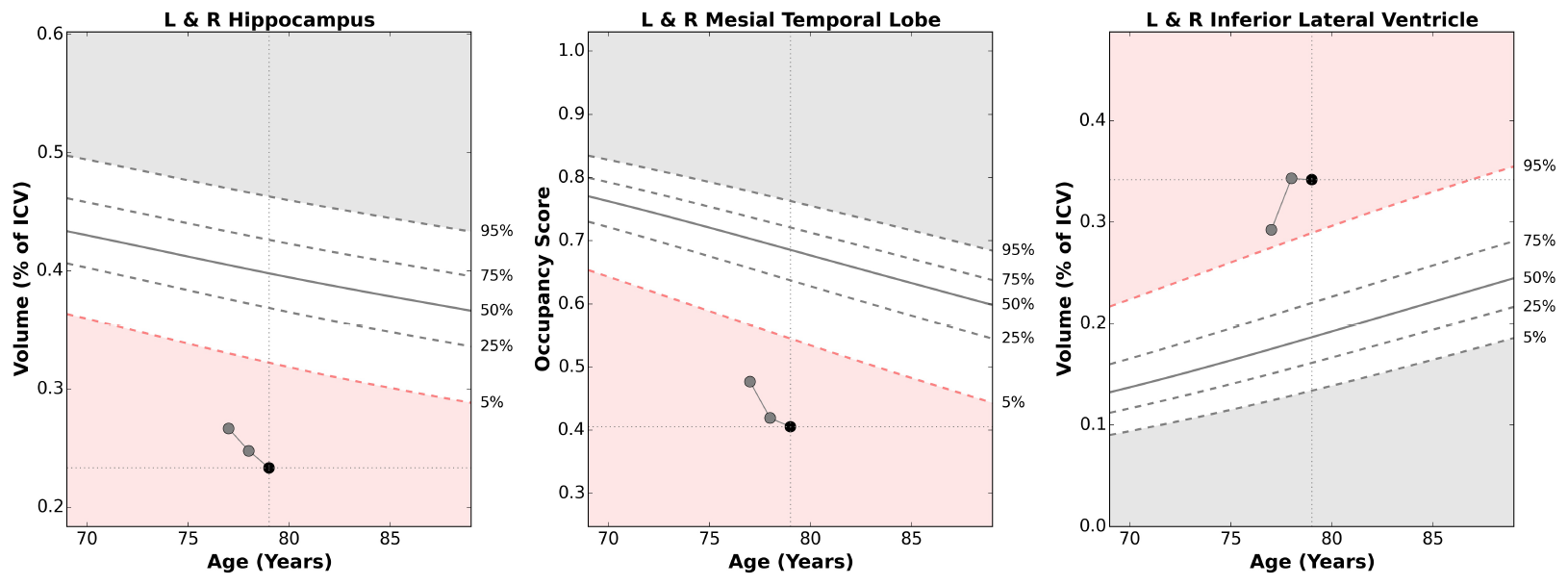
The chart below depicts the same patient two years later at 79 years old. NeuroQuant provides the same three brain structures measurements as before. The patient has had annual scans resulting in three data points for each brain structure. The visualization in one plot indicates the progression of the atrophy over time, and a physician can view the worsening hippocampal atrophy.
NeuroQuant provides the longitudinal reporting information in a single NeuroQuant volumetric output report. When multiple time point measurements of the same patient are available, the results are displayed on a single report, allowing physicians to see a clear progression of any changes to the patient’s relevant brain structures. The use of a single report allows the physician to quickly and easily evaluate any trend in the patient’s brain volume development.
NeuroQuant’s longitudinal reporting feature is a major component in the personalized care path that follows an initial evaluation. It provides physicians with more objective data to help with clinical assessments and when outlining possible clinical courses.