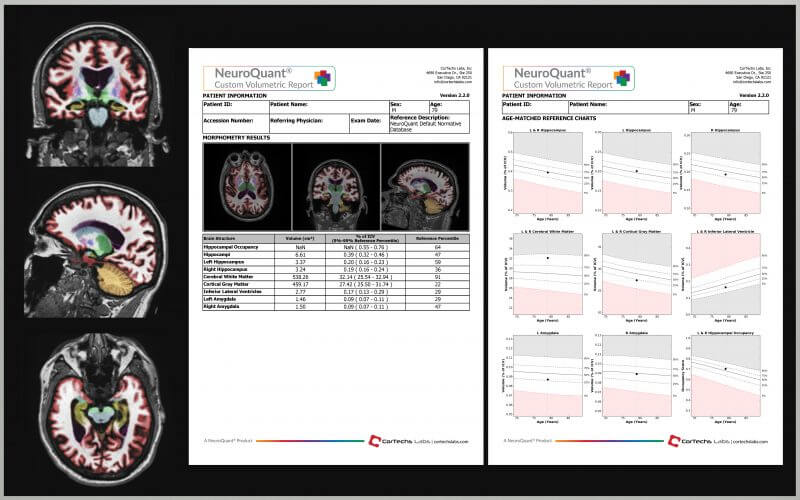
In mid-October, we announced the latest software release of NeuroQuant. This latest version, based almost entirely on customer feedback, adds exciting new features to the current version of NeuroQuant, including the ability for NeuroQuant users to customize their own volumetric reports.
The NeuroQuant Custom Volumetric report allows physicians to individually tailor NeuroQuant reports relevant to their clinical cases. Users are able to choose between left, right, total brain structure volume, as well as left-right asymmetry for up to 9 different brains structures per report.
Features and Benefits of the Custom Volumetric Report
- Select from 71 available brain structures
- Reduce the overall number of standard NeuroQuant reports to review
- Evaluate for neurological conditions not available in standard NeuroQuant reports
- Incorporate brain structures not displayed in standard NeuroQuant reports
- Compare results to age and gender-matched reference data or alternative (MCI or AD) reference data (alternative reference data is not for clinical use)
Watch our latest webinar about Customizing NeuroQuant Reports and Recent Software Updates here.
A separate NeuroQuant license is required to use the Custom report feature, so contact your sales representative today!