San Diego, CA, April. 22, 2025 – Cortechs.ai, a global leader in AI-driven medical imaging analysis, is proud to announce that that it has received approval from Health Canada for the sale of its innovative suite of imaging solutions. This regulatory milestone marks a significant expansion of Cortechs.ai’s presence in the Canadian healthcare market and reinforces its commitment to enhancing clinical diagnostics and radiology workflows worldwide.
Cortechs.ai’s advanced portfolio includes NeuroQuant®, NeuroQuant® MS, OnQ™ Prostate, and NeuroQuant® Brain Tumor. These software tools harness advanced AI to streamline complex imaging analysis, equipping clinicians with accurate insights that enhance disease detection and validation across various care pathways.
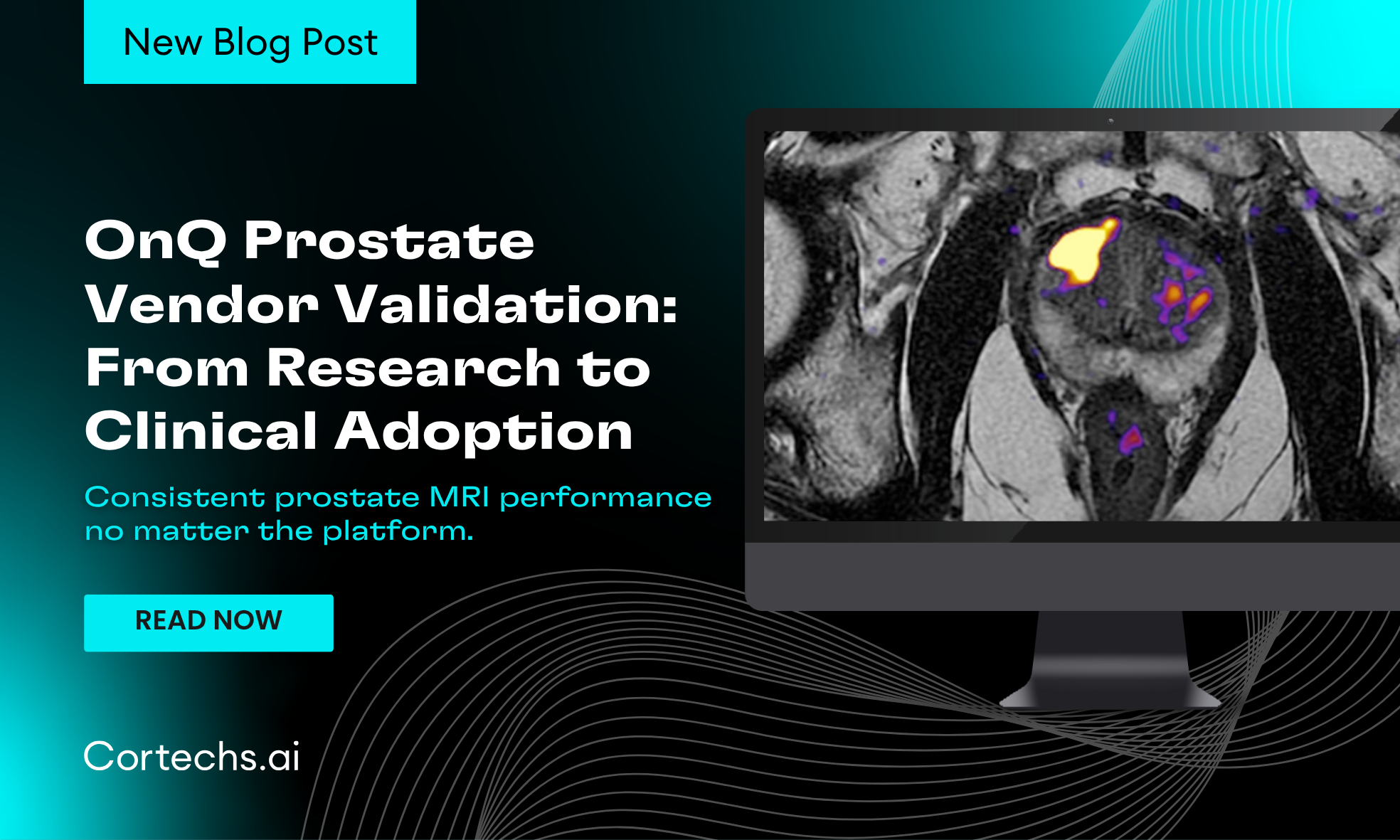
NeuroQuant® and NeuroQuant® MS offer automated brain segmentation and volumetric analysis, aiding in the early detection and monitoring of neurodegenerative diseases such as Alzheimer’s and Multiple Sclerosis. NeuroQuant® Brain Tumor delivers quantitative imaging analysis to assist clinicians in evaluating brain tumor progression, while OnQ™ Prostate enhances prostate MRI interpretation, enabling more precise detection and assessment of prostate cancer.
“This Health Canada approval is a monumental achievement for Cortechs.ai and a testament to our dedication to transforming diagnostic imaging,” said Jared Dixon, VP of Global Sales at Cortechs.ai. “Expanding access to our AI-powered solutions in Canada means that more clinicians will have the tools they need to make faster, more accurate diagnoses and improve patient outcomes. This is a major step forward for our company’s growth and our mission to revolutionize disease detection and treatment across the globe.”
With this approval, Cortechs.ai is poised to collaborate with healthcare providers and institutions across Canada, equipping them with cutting-edge solutions that optimize clinical workflows and elevate patient care.
For more information about Cortechs.ai and its cutting-edge imaging solutions, visit www.cortechs.ai.
About Cortechs.ai
Cortechs.ai is a leader in AI applications in radiology. Cortechs.ai uses cutting-edge technologies in medical imaging to revolutionize disease screening and early detection so patients can enjoy longer, healthier lives. The company develops and markets breakthrough medical device software that quantifies and tracks neurodegenerative diseases, and assists in the detection of clinically significant cancer. Cortechs.ai’s industry-leading brain imaging software provides radiologists, neurologists, oncologists, and clinical researchers worldwide with a convenient and cost-effective way to quantify brain structures for assessing Alzheimer’s disease, epilepsy, multiple sclerosis, brain trauma, and other brain abnormalities. The company has FDA-cleared products for use in helping the diagnosis and follow-up of neurodegenerative and traumatic brain conditions, as well as prostate cancer. Please visit www.cortechs.ai for further information and follow us on Twitter, LinkedIn, and Facebook.
For more information, please contact:
___________
info@cortechs.ai
+1 858 459 9700