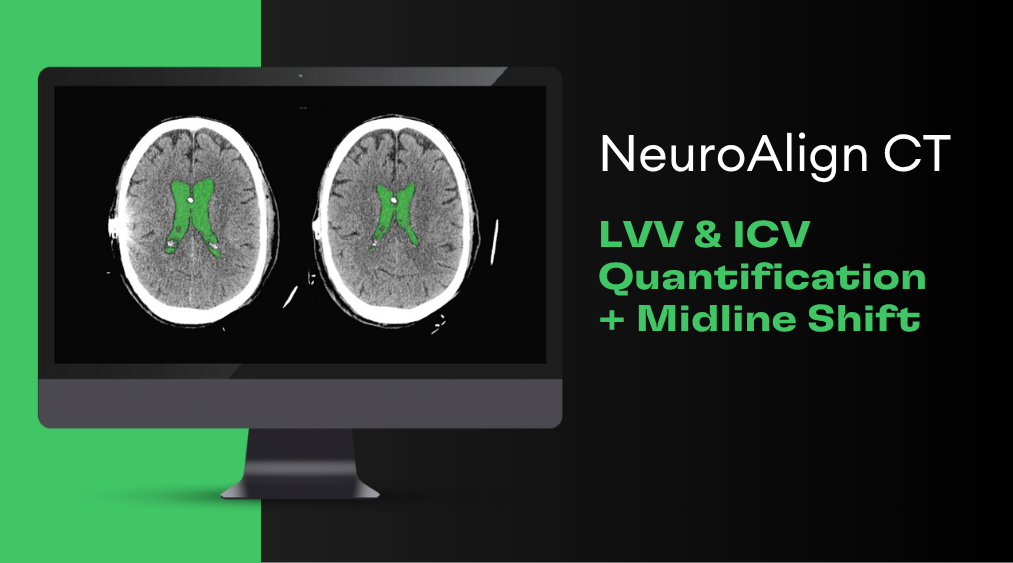
Phase two of this grant proves Cortechs.ai’ PETQuant™ to be clinically and commercially viable
(SAN DIEGO) November 7, 2019 – Cortechs.ai Inc. announced today that it has officially entered the second phase of its project titled, Diagnosis of Alzheimer’s disease and prediction of clinical progression using an automated PET image analysis tool. This project, funded by a $1.15 million, multi-year grant from the National Institutes of Health (NIH), was awarded to Cortechs.ai in December of 2018.
The grant was awarded to reshape how Alzheimer’s disease is diagnosed. During the first phase, Cortechs.ai worked on delivery methods using an automated PET image analysis tool to determine a patient’s risk of developing Alzheimer’s and to monitor disease progression and treatment response.
In the second phase, Cortechs.ai is developing an automatic classification algorithm with the goal of separating Alzheimer’s disease patients from non-Alzheimer’s disease controls. The company will also assess if the algorithm can predict the risk of developing the disease in individuals. Cortechs.ai is establishing normative values to effectively identify scans that stray from the typical population. This entails carefully examining the degree of cortical binding of amyloid agents so that physicians can distinguish Alzheimer’s disease from diseases with similar manifestations, such as Lewy body dementia or cerebral amyloid angiopathy.
“PETQuant yields a remarkable advance in the interpretation of PET scans, providing physicians with objective quantification of tracer binding in native brain space,” said Dr. James Brewer, Director of the Shiley-Marcos Alzheimer’s Disease Research Center at the University of California, San Diego. “Faster and more consistent reads will accelerate the adoption of PET imaging to provide better and faster assessment and classification of dementia patients.”
Alzheimer’s disease is the sixth leading cause of death in the United States and is becoming one of the biggest health battles of today. The Alzheimer’s Association states that Alzheimer’s is a progressive disease, where dementia symptoms gradually worsen over a number of years. In its early stages, memory loss is mild, but with late-stage Alzheimer’s, individuals lose the ability to carry on a conversation and respond to their environment. It is important to find ways to diagnose as early as possible and to tap the potential of modern, cutting-edge technology.
“Our goal is to provide a better, more accurate diagnosis of Alzheimer’s disease to aid in the fight against this devastating illness,” said Chris Airriess, chief executive officer of Cortechs.ai. “This work will not only deepen our understanding of the effects of Alzheimer’s on the brain to aid in early, accurate diagnosis, but will also move PETQuant, a research-specific component of NeuroQuant® that aids in the study of frontotemporal dementia and Alzheimer’s disease, from a research tool to a clinical tool as we progress towards regulatory clearance in various jurisdictions.”
About Cortechs.ai
Cortechs.ai develops and markets breakthrough medical device software solutions capable of automatically segmenting and quantifying brain structures, making quantitative analysis of the human brain a routine part of clinical practice. Cortechs.ai’ cutting-edge brain imaging analysis provides neurologists, radiologists and clinical researchers worldwide with a convenient and cost-effective means to quantify brain structures to help assess a variety of neurological conditions, such as Alzheimer’s disease, epilepsy, multiple sclerosis, brain trauma and brain development abnormalities. Please visit cortechs.ai for further information and follow us on Twitter, LinkedIn and Facebook.
###