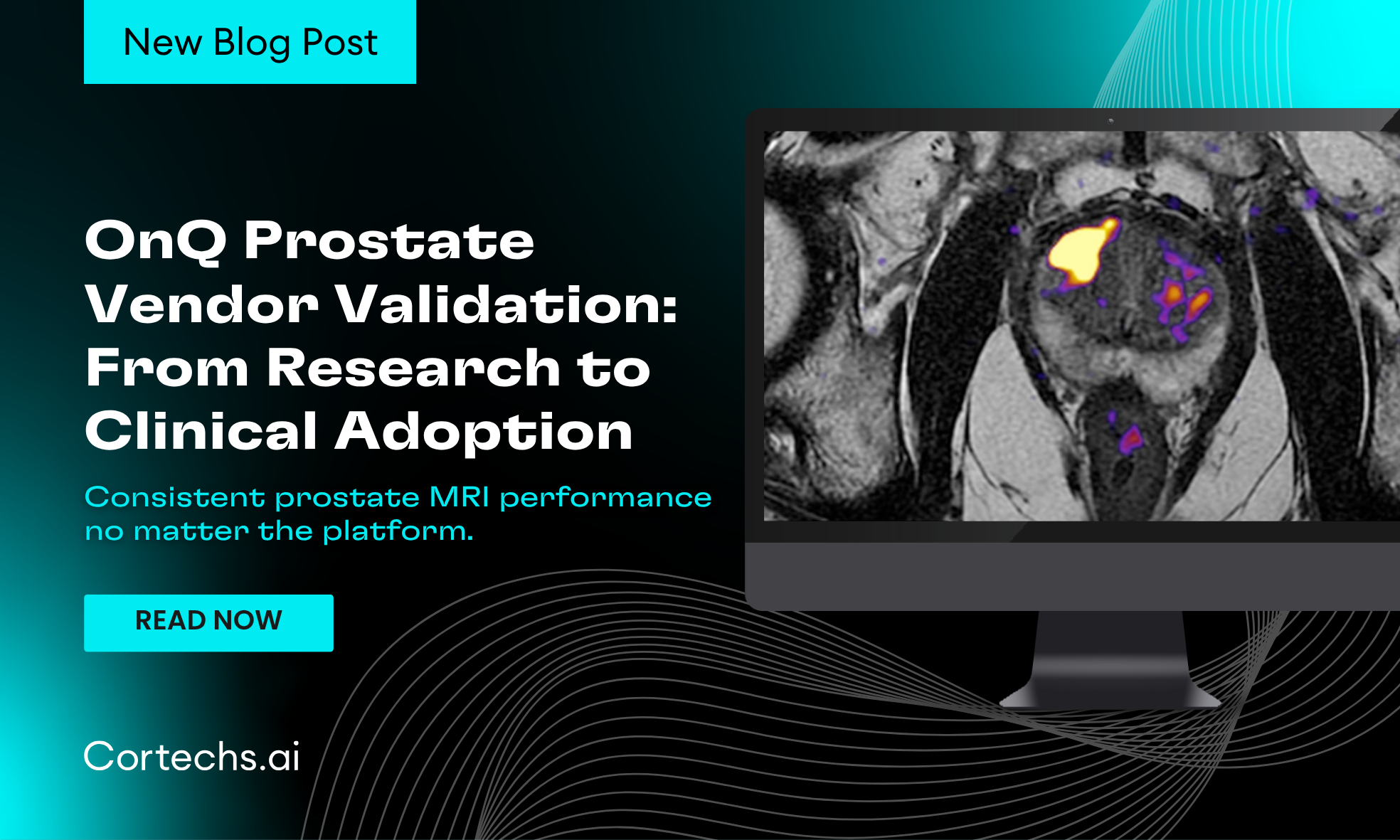
Prostate MRI has become an essential tool in prostate cancer detection and treatment planning. One of the greatest challenges in clinical imaging today is variability-differences in MRI platforms, field strengths, and acquisition approaches can influence image quality and diagnostic confidence. OnQ Prostate was initially implemented on GE and Siemens 3T systems in a research setting. With Cortechs.ai’s commitment to delivering clinically ready AI solution we have expanded to include Fuji, Canon, United, and Philips, on both 1.5T and 3T systems.
At Cortechs.ai, we believe advanced AI solutions must deliver consistent, reliable performance regardless of where or how they are deployed. That’s why vendor validation is a foundational element of OnQ Prostate.
Why Vendor Validation Matters
Vendor validation helps ensure that OnQ Prostate delivers dependable performance by confirming that:
- Quantitative outputs remain reliable across supported MRI vendor platforms
- Qualitative outputs are intuitive and clinically meaningful
- Reports are clear, consistent, and suitable for routine clinical use and reimbursement
For AI-powered prostate imaging, vendor validation is not optional, it is essential for clinical trust and scalability.
Designed for Real-World, Multi‑Vendor Imaging
OnQ Prostate is built with real-world clinical environments in mind. Vendor validation testing confirms that when supported MRI input such as T2-weighted imaging and RSI diffusion-weighted imaging are provided, the software consistently generates high-quality, clinically usable outputs across vendor platforms.
Throughout validation testing, OnQ Prostate demonstrated reliable performance in generating:
- Restricted Signal Maps
- Color Fusion Maps
- ADC and Trace-Weighted Images
- Quantitative Prostate Reports
Clinical reviewers consistently found these outputs to be intuitive, legible, and easy to interpret, supporting efficient and confident prostate MRI review across imaging environments.
Supporting Clinical Confidence at Scale
Vendor validation helps ensure that OnQ Prostate delivers dependable qualitative and quantitative insight-even in diverse, multi‑vendor imaging environments. By reducing technical uncertainty, radiologists can focus on what matters most: interpretation, diagnosis, and patient care.
With OnQ Prostate, imaging teams benefit from:
- Confidence in prostate MRI results especially diffusion weighted imaging
- Consistent visualization across supported MRI vendors
- A streamlined workflow that integrates seamlessly into everyday clinical practice
Advancing Quantified Prostate MRI
OnQ Prostate vendor validation reflects Cortechs.ai’s commitment to delivering clinically ready AI solutions, designed, tested, and validated for real-world use. Through rigorous vendor validation, OnQ Prostate demonstrates its readiness to support advanced prostate MRI across imaging platforms, helping clinicians move forward with confidence.