What technology is used in OnQ® Prostate?
OnQ Prostate uses Restriction Spectrum Imaging (RSI), an advanced diffusion-weighted magnetic resonance imaging (MRI) technique, to “highlight” areas within the prostate that have the cellular characteristics of cancer. Through the use of a sophisticated, multi-compartment tissue microstructure model, RSI is able to provide a voxel-level classifier of prostate cancer that is superior to conventional imaging.[1]
How does OnQ Prostate improve prostate MRI?
Current diffusion-weighted imaging (DWI) used in multiparametric(mp) MRI has inherent limitations for prostate cancer detection because it is susceptible to a number of factors that impact sensitivity and specificity.[2] The clinical consequences include overdiagnosis of benign or indolent disease, underdiagnosis of some clinically significant cancers, and interobserver variability amongst radiologists. OnQ Prostate was developed to optimize visualization and detection of tumor cells, directly addressing the shortcomings of standard DWI for oncology.[3] Resultantly, OnQ Prostate improves detection accuracy for clinically significant prostate cancer and reduces interobserver variability compared to conventional mpMRI.[4] Patient benefits may include avoidance of unnecessary biopsies or more accurately targeted biopsies and treatments for overall better outcomes and experience.
Which patients are suitable for OnQ Prostate?
OnQ Prostate is appropriate for all patients undergoing prostate MRI. Current evidence supports MRI in biopsy-naive men at risk for prostate cancer and men who have increasing PSA levels after an initial negative biopsy.[5] OnQ Prostate has recently shown to significantly improve prostate cancer imaging to predict upgrading in men with low-risk disease who are on active surveillance.[6]
How is OnQ Prostate used in clinical practice?
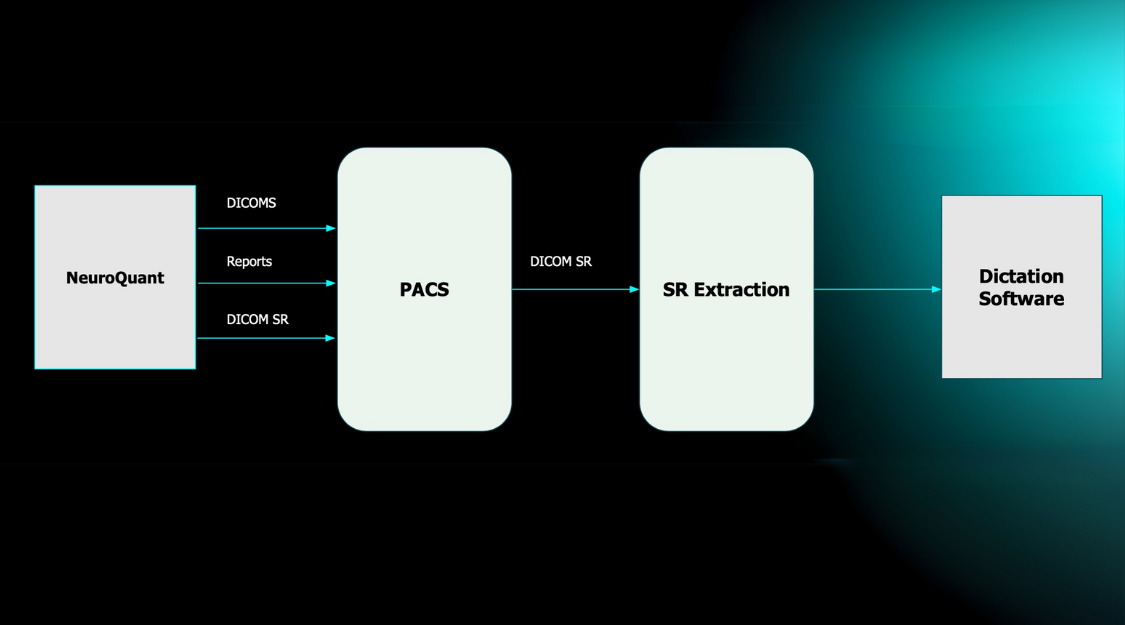
OnQ Prostate is typically performed in combination with standard prostate mpMRI. Once the patient is scanned, MRI data are processed automatically and output images are returned directly to PACS, with no change in workflow or separate workstation required. OnQ Prostate makes suspicious areas easier to see and harder to miss, enabling faster, more accurate PI-RADS scoring. These images can be shared with referring urologists and radiation oncologists to assist with targeted biopsy and treatment decision-making and procedure planning.
Click here to watch a case study of OnQ Prostate in clinical practice at Imaging Health Specialists.
What research supports the technology used by OnQ Prostate?
The technique used by OnQ Prostate has been established in the literature as an evolving imaging biomarker for clinically significant prostate cancer since 2016.[7] Numerous studies have shown that added to mpMRI, OnQ Prostate’s technology improves the detection of clinically significant prostate cancer compared to mpMRI alone. Furthermore, this method performed alone with T2-weighted imaging achieves equivalent detection performance to mpMRI, important for patients who cannot receive contrast and practices looking to implement biparametric MRI.[4]
References
[1] https://doi.org/10.1002/jmri.27623
[2] https://doi.org/10.1007/s00330-010-1960-y
[3] https://doi.org/10.1158/0008-5472.CAN-13-3534
[4 ] https://doi.org/10.1007/s00261-016-0659-1
[5] https://doi.org/10.1016/S0140-6736(16)32401-1
[6] https://doi.org/10.1097/JU.0000000000001692
[7] https://doi.org/10.1002/jmri.25419