Dr. Ross Schwartzberg | Imaging Healthcare Specialists
With over 30 years of imaging experience, Imaging Healthcare Specialists has earned an outstanding reputation for providing the highest quality medical imaging technology, highly specialized expertise, and exceptional customer service to physicians and patients. The prostate MRI program initiated by Dr. Ross Schwartzberg in 2013, has experienced progressive growth and is expected to perform approximately 1400 prostate MRI exams and 120 MRI in-bore targeted biopsies in 2021. Imaging Healthcare Specialists partners with Cortechs.ai to provide the latest advancement in diffusion MRI by integrating OnQ Prostate to support improved detection of clinically significant prostate in patients.

OnQ Prostate Overview
OnQ Prostate is an FDA-cleared post-processing application from Cortechs.ai that supports improved detection of clinically significant prostate cancer. OnQ Prostate leverages an advanced diffusion MRI technique called Restriction Spectrum Imaging (RSI), powered by AI, to enable faster, more accurate PI-RADS scoring. Compared to conventional MRI alone, RSI has demonstrated superior accuracy for discriminating between aggressive prostate cancer (PCa) and normal prostate tissue, and improved correlation with Gleason Score. RSI with multiparametric (mp)MRI improves PCa detection compared to mpMRI alone, while biparametric (bp)MRI with only RSI and T2-weighted imaging achieves equivalent performance to mpMRI. In addition, RSI can improve inter-reader agreement by improving consistency across radiologists.
OnQ Prostate at Imaging Healthcare Specialists
Imaging Healthcare Specialists was the first outpatient imaging provider to adopt OnQ Prostate as part of all clinical routine prostate MRI exams, and Dr. Schwartzberg quickly became a champion of the technology. Dr. Schwartzberg testifies to the utility of OnQ Prostate for PI-RADS scoring, stating that it allows him to more quickly and confidently rule in or rule out the likely presence of clinically significant prostate cancer. This allows many men to safely avoid a biopsy and provides more accurate targeting for biopsies that are deemed necessary by MRI. As an expert radiologist, Dr. Schwartzberg appreciates the added efficiency and clinical confidence that OnQ Prostate provides in typical routine exams, along with the needed assistance in more complex cases. He also acknowledges that OnQ Prostate has the potential to “level the playing field” for less experienced or less specialized readers to improve overall accuracy and performance.
Case Examples
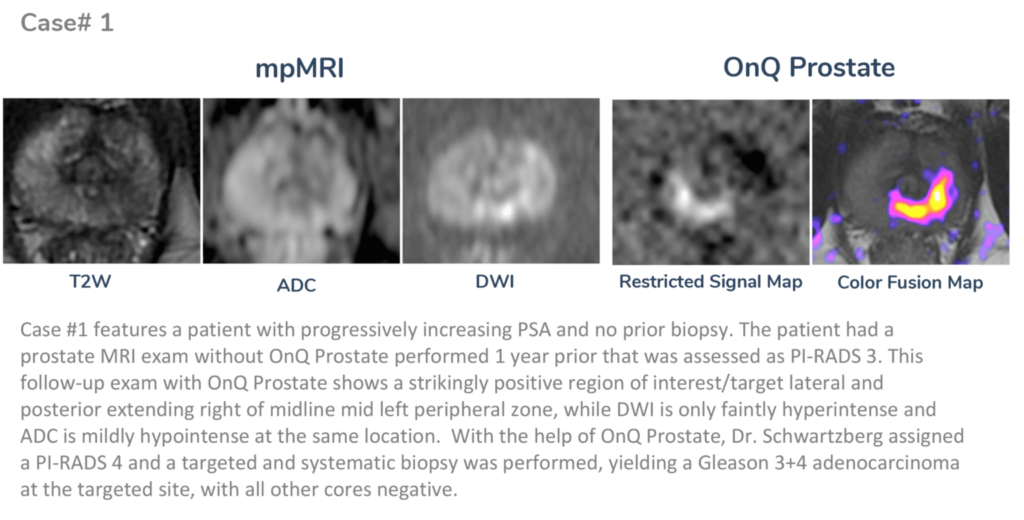
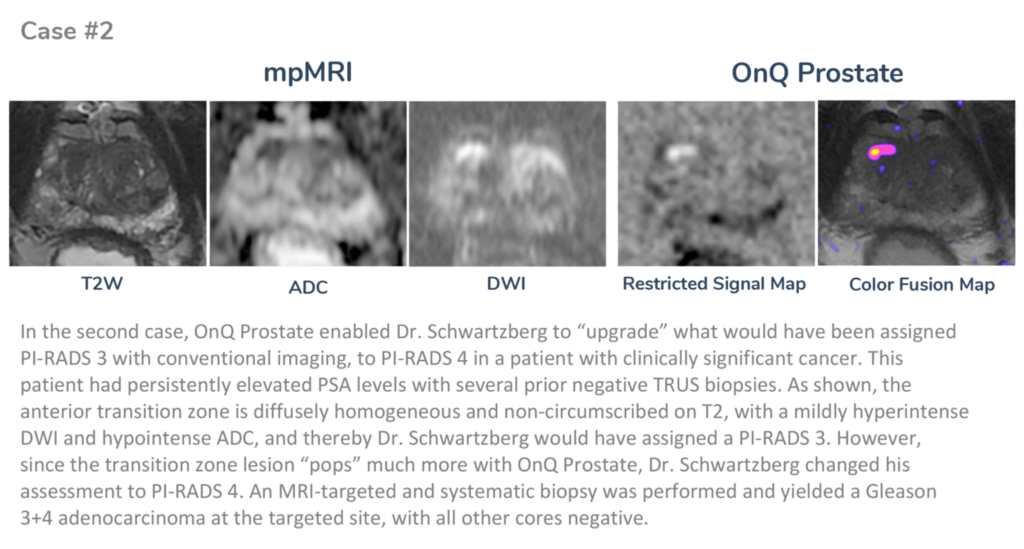

About Dr. Schwartzberg
Ross E. Schwartzberg, M.D. is a board-certified Neuroradiologist. He earned his medical degree from the University of Arizona College of Medicine in Tucson, Arizona. An internship in Internal Medicine at St. Mary’s Hospital in San Francisco brought him to the Bay Area. He then completed a Radiology residency and a two-year fellowship training program in Neuroradiology at Stanford University Hospital and Medical Center.







