By Brandon Steach
Traumatic brain injury (TBI) occurs when a sudden, external, physical assault causes damage to the brain. This severity of the trauma can range from a minor blow to the head to a severe penetrating mortal injury. In recent years, the world has become more cognizant of mild TBI (mTBI) aka the “brain concussion”. TBI can have a variety of adverse neuropsychological outcomes. The clinical outcome depends on many different factors. The evaluation and classification of the severity requires an astute medical professional, or preferably, a team of subspecialized professionals that consider all the different medical testing, reported symptoms, specialist consultations, and physiological variables involved with the brain injury.
One of the staple tests for a thorough evaluation is an MRI. The 3T MRI protocol recommendations from Transforming Research and Clinical Knowledge in Traumatic Brain Injury, (TRACK-TBI) an international traumatic brain injury research initiative, are: a 3D T1 gradient echo (GRE) for volumetric and morphometry, a 3D T2* GRE to detect microbleeds, diffusion tensor imaging (DTI), resting state BOLD fMRI to evaluate functional connectivity, 3D T2 FLAIR to detect lesions, and a 3D T2 for volumetrics and morphometry.
Some of these recommended sequences may not be available on the real world commercial / clinical equipment currently in use. However, the 3D T1 gradient echo is a common sequence available on most 1.5T and 3T MRIs. This sequence has been an essential for its high resolution anatomical detail that is used for surgical planning, radiotherapy, and volumetric analysis.
Advancements in artificial intelligence neuroimaging software technology have utilized this 3D T1 sequence to extrapolate unbiased volumes to age and gender match controlled normative samples. This is our specialty at Cortechs.ai and we have a quantitative MRI (QMRI) product specifically to address TBI.
Our updated version 4.0 of NeuroQuant® Triage Brain Atrophy (TBA) can be used for a variety of neurodegenerative conditions including: brain trauma, post-traumatic stress disorder (PTSD), diffuse axonal injury (DAI), and chronic traumatic encephalopathy (CTE).
NeuroQuant® TBA measures 33 different pertinent brain structures related to TBI, and the new release has simplified the report output to prioritize the order of structures, so that more commonly affected areas of TBI are listed first. Less relevant structures were removed on version 4.0.
The color coded key was simplified to one color in order to enhance visualization of abnormal values (structures below the 5th percentile or a ventricle above the 95th percentile) to identify important biomarkers more easily (see red arrow in 4.0 report example).
The segmentation colors have been enriched between versions to improve visualization. Our NeuroQuant® algorithm is now a hybrid deep learning (DL) and machine learning (ML) model. The new model significantly improved cortical gray matter segmentation, which may result in minimal variation in reported percentiles, when compared to earlier software versions.
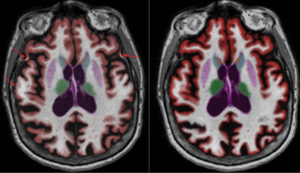
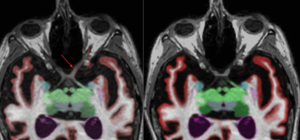
The improved accuracy of the new model is also shown in many other areas of over-segmentation, as can be seen in the optic nerve below. Take a look at the case that was processed on version 3.1.2 of NeuroQuant, versus a case that was processed on version 4.0 with the updated segmentation model.
Example 1:
Example 2:
TBI and volumetric findings have been researched for decades and there has been documented abnormal asymmetry of brain regions especially with severe TBI cases (1-3). In regard to chronic mild to moderate TBI the findings can be more difficult to evaluate due to the pathophysiology and mechanism of injury. In these instances, it has been documented that some patients have atrophy of cerebral white matter and a few other regions, but they had even more regions of abnormal enlargement, including cortical gray matter regions and subcortical regions (4-8). These areas of abnormal enlargement have been hypothesized to be a result of a compensatory response to more-injured regions of the brain (9).
In a published case study (9) of a patient with chronic moderate traumatic brain injury the atrophy and abnormal asymmetry of the cerebral white matter correlated with bradyphrenia and executive dysfunction (10, 11). The asymmetrical thalamus corresponded with the reported impaired sleep and wakefulness (12). The increased volume of the posterior cingulate gyri correlated with impaired mood and visuospatial skills (13, 14). The abnormal asymmetry and enlargement of the right temporal pole was linked to the impaired mood (15) and impaired emotional empathy/irritability (16). Other areas of enlargement that corresponded to the patient’s symptoms were the transverse temporal, superior temporal, and the hippocampi which was reported asymmetric as well.
The NeuroQuant TBA Report provides physicians with an assessment of 47 regional + global brain structure volume measurements for patients with a history of traumatic brain injury, and help assist in evaluating the extent or impact of TBI – allowing improvement in patient care.
References:
- Bigler ED. Structural imaging. In: Textbook of Traumatic Brain Injury. Silver JM, Mcallister TW, Yudofsky SC (Eds). American Psychiatric Publishing, Inc, DC, USA, 79–105 (2005).
- Bigler ED. Structural imaging. In: Textbook of Traumatic Brain Injury. Silver JM, Mcallister TW, Yudofsky SC (Eds). American Psychiatric Publishing, Inc, DC, USA, 73–90 (2011).
- Wilde EA, Little D. Clinical Imaging. In: Textbook of Traumatic Brain Injury. Silver JM, Mcallister TW, Arciniegas DB (Eds). American Psychiatric Association Publishing, DC, USA, 89– 126 (2019).
- Ross DE, Ochs AL, Zannoni MD, Seabaugh JM. Back to the future: estimating pre-injury brain volume in patients with traumatic brain injury. Neuroimage 102, 565–578 (2014).
- Wang X, Xie H, Cotton A et al. Early cortical thickness change after mild traumatic brain injury following motor vehicle collision. J. Neurotrauma 32(7), 455–463 (2015).
- Govindarajan KA, Narayana PA, Hasan KM et al. Cortical thickness in mild traumatic brain injury. J. Neurotrauma 33(20), 1809–1817 (2016).
- Ross DE, Ochs AL, Zannoni MD, Seabaugh JM. Corrigendum to “Back to the Future: estimating pre-injury brain volume in patients with traumatic brain injury” [NeuroImage 102 (Part 2) (15 November 2014) 565–578]. Neuroimage 127, 510–511(2016).
- Ross DE, Seabaugh JD, Seabaugh JM et al. Patients with chronic mild or moderate traumatic brain injury have abnormal brain enlargement. Brain Inj. 34, 11–19 (2020).
- Barcelona J, Ross, D, Seabaugh J, Seabaugh J. Abnormal asymmetry correlates with abnormal enlargement in a patient with chronic moderate traumatic brain injury. Concussion 7(1), (2022) https://doi.org/10.2217/cnc-2021-0006
- Ylikoski R, Ylikoski A, Erkinjuntti T, Sulkava R, Raininko R, Tilvis R. White matter changes in healthy elderly persons correlate with attention and speed of mental processing. Arch. Neurol. 50(8), 818–824 (1993).
- Alosco M, Koerte I, Tripodis Y et al. White matter signal abnormalities in former national football league players. Neuroimaging10, 56–65 (2018).
- Steriade M, Llinas RR. The functional states of the thalamus and the associated neuronal interplay. Physiol. Rev. 68, 649–742 (1988).
- Maddock R, Garrett A, Buonocore M. Rembering familiar people: the posterior cingulate cortex and autobiographical memoryretrieval. Neuroscience 104(3), 667–676 (2001).
- Cavanna A, Trimble M. The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129(3), 564–583 (2006).